Introduction
Our human ancestors evolved and developed adaptive mechanisms that allowed survival during periods of food availability and scarcity with seasonal fluctuations of both types and quantities of nutrients.1 In this context, modern humans now face a different challenge with increased food abundance and food accessibility. Not only has the number of commercial food establishments increased dramatically in the last few decades, a large percentage of nonfood establishments (e.g., retail outlets, gas stations, office buildings, etc.) also have made food readily available.2 Further, the increase in food and beverage portion sizes and the ubiquitous food advertising lead people to unintentionally overconsume calories.3,4 The resulting epidemic of obesity, diabetes, metabolic dysfunction, and associated conditions is leading healthcare professionals and researchers into a quest for approaches to mitigate these adverse health outcomes.
Continuous energy restriction (CER), or traditional dieting, is a method to increase metabolic health by consistently restricting daily caloric intake (reducing 20-40% of caloric intake) without malnutrition or deprivation of essential nutrients.5 However, practicing CER while increasing energy expenditure via physical activity can be challenging to adhere to. Even when weight loss is achieved, weight regain eventually occurs to many people due to adaptive physiological changes following weight loss.6 Intermittent fasting (IF), also commonly known as intermittent energy restriction (IER), may be an alternative, potentially more agreeable, approach to reduce energy intake.7
| Download the PDF |
Mechanisms of action
An intermittent energy deficit via dietary restriction can be considered a hormetic stimulus, defined as a moderate stressor that results in adaptive responses leading to increased activation of cellular protection and restorative mechanisms.8 The proposed mechanisms via which IF may lead to improved health outcomes are believed to be linked to IF’s effects on multiple, interrelated cell signaling pathways:9
- Activation of more efficient respiratory pathways such as AMP-dependent kinase (AMPK) and sirtuins leads to improved mitochondrial efficiency and reduced production of reactive oxygen species (ROS)
- Activation of the Forkhead box (FoxO) pathway facilitates autophagy (a process in which cells degrade intracellular debris, thereby eliminating dysfunctional proteins and organelles)
- Downregulation of insulin-like growth factor-1 (IGF-1) receptor-dependent pathways and target of rapamycin (TOR)-dependent activities results in inhibition in cell proliferation and glycolysis
- Inhibition of nuclear factor-κB (NF-κB) activity produces anti-inflammatory effects
- Activation of the nuclear factor-like 2 (Nrf2) pathway increases the expression of antioxidant enzymes
Research Highlights
- Energy restriction consists of periods with limited or no caloric intakes that results in adaptive mechanisms to support homeostasis.8,9
- Energy restriction/fasting protocols are associated with beneficial metabolic effects such as reduced blood levels of glucose and insulin, improved insulin sensitivity, and beneficial changes in body weight and body composition.10
- Energy restriction can be achieved via continuous energy restriction (CER) and various methods of intermittent fasting (IF). IF may be an alternative, potentially more agreeable, approach to reduce energy intake.7
Different IF approaches
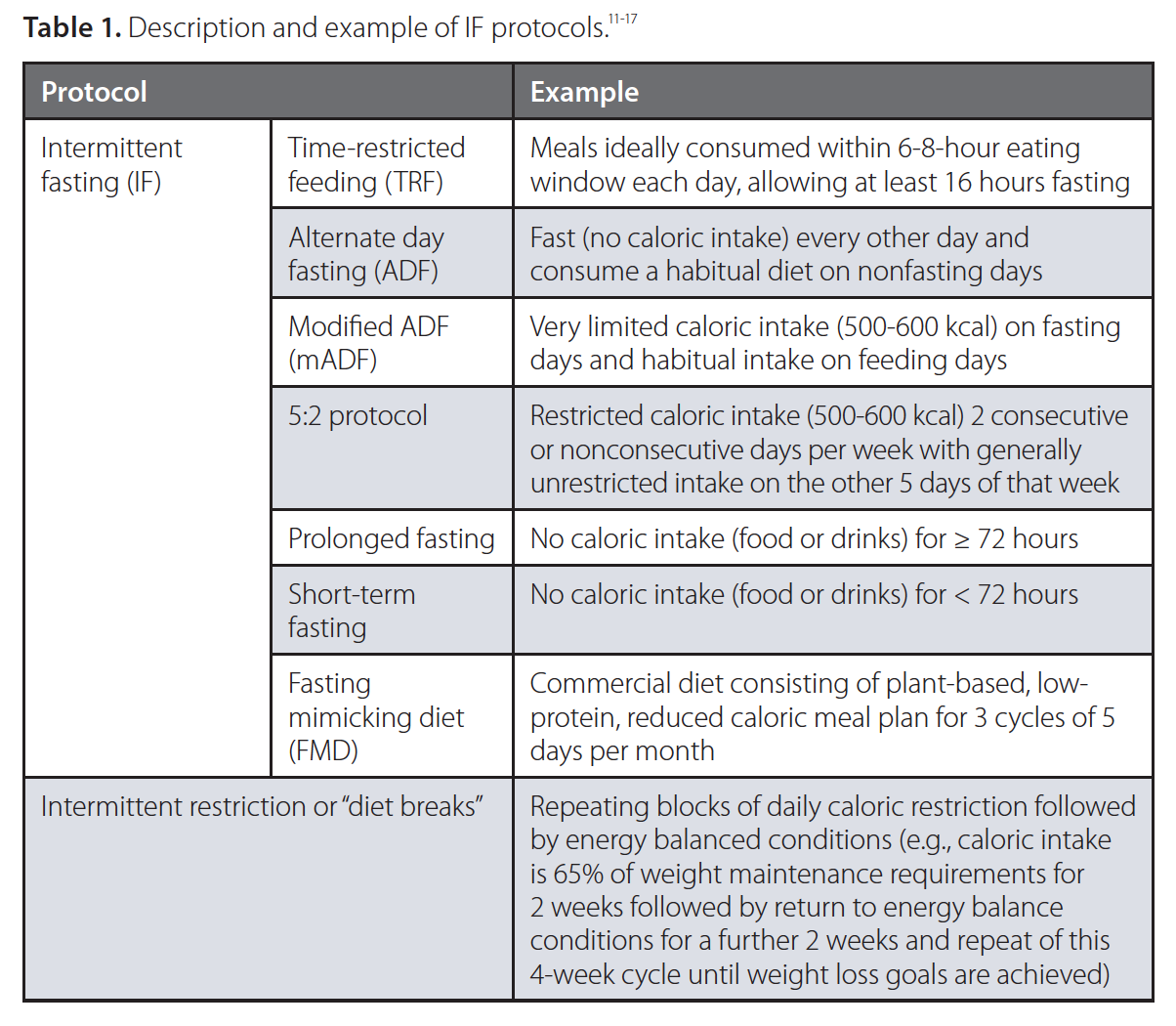
IF can be achieved via different protocols described in Table 1.
 Reduced nutrient intake from IF may result in compensatory metabolic effects such as:10
Reduced nutrient intake from IF may result in compensatory metabolic effects such as:10
- Reduced blood levels of glucose and insulin together with hepatic glycogen depletion
- Improved insulin sensitivity, increased fat oxidation, and beneficial changes in body composition
- Improved cellular utilization of ketones and fatty acids and beneficial changes in lipid profile (e.g., decreased total cholesterol, triglycerides, and LDL cholesterol and increased HDL cholesterol levels)
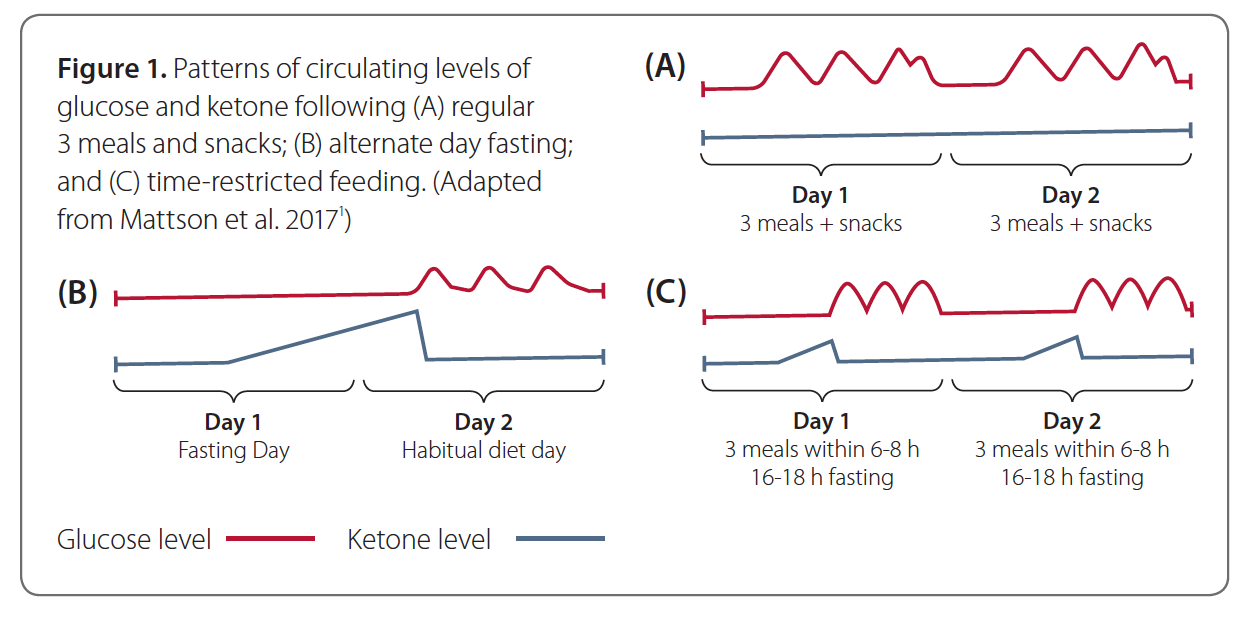
However, different IF protocols have been shown to produce subtle differences in positive metabolic changes. For instance, ADF leads to a gradual and lengthier increase in ketone levels on fasting day until the first consumption on the second day, whereas TRF results in an increase in ketone levels during the last 6-8 hours of the 16-18-hour fasting time (Figure 1).1
Potential clinical applications
Investigating long-term energy restriction in humans can be challenging. The first long-term human energy restriction clinical trial is the Comprehensive Assessment of Long-term Effects of Reducing Intake of Energy (CALERIE) trial, which found that a 12% reduction in caloric intake for up to 2 years reduced risk for age-related diseases such as diabetes, heart disease, and stroke.18,19
Due to its multiple effects, IF protocols may have several potential clinical applications such as weight management, diabetes management, cardiometabolic improvement, and cognitive and brain health support (evidence from key human clinical trials and/or preclinical studies for each application is briefly summarized in Table 2 along with references).
Choosing between IF and energy restriction protocols
At this point, head-to-head studies of different IF and energy restriction protocols are lacking. However, it is possible to differentiate between protocols based on emerging clinical applications as well as individual practical lifestyle aspects.
Practicality and safety of IF
Investigating long-term energy restriction in humans can be challenging. The first long-term human energy restriction clinical trial is the Comprehensive Assessment of Long-term Effects of Reducing Intake of Energy (CALERIE) trial, which found that a 12% reduction in caloric intake for up to 2 years reduced risk for age-related diseases such as diabetes, heart disease, and stroke.18,19 Due to its multiple effects, IF protocols may have several potential clinical applications such as weight management, diabetes management, cardiometabolic improvement, and cognitive and brain health support (evidence from key human clinical trials and/or preclinical studies for each application is briefly summarized in Table 2 along with references).
Fast day meal timing may also be flexible to increase tolerability and long-term adherence to ADF or mADF protocols. One trial reported that eating the fast day meal at lunch time, moving the fast day meal to dinnertime, or dividing the fast day meal into smaller meals were equally effective in improving body weight and body composition in adults with obesity over 8 weeks.22
Exercise combined with IF may lead to greater weight loss than exercise alone or IF alone.23 One trial found that exercise could be easily incorporated into the ADF regimen either on fast days or feed days, and exercise on fast days did not result in increased hunger or extra food intake in subjects with obesity over 12 weeks.24
For people using type 2 diabetes (T2D) medication, IF may require medication changes and regular monitoring—especially in the initial stages of dietary change—to prevent hypoglycemia.25 One trial reported that sulfonylureas and insulin dosage needed to be adjusted on fasting days according to participants’ baseline HbA1c levels (e.g., discontinuing sulfonylureas and insulin on fasting days only and discontinuing long-acting insulin the night before a fasting day for those with HbA1c levels between 7 and 10%) and recommended that patients work with endocrinologists while adapting to IF regimens.25
For people with type 1 diabetes (T1D) and obesity, a 12-week pilot trial demonstrated IF (as 5:2 protocol) to be a safe and effective alternative to traditional CER for weight loss, and moderate adjustment in their basal insulin doses (10% reduction on nonfasting days and 50% reduction on fasting day) effectively prevented hypoglycemia.26
Fasting protocols have not been investigated in children, elderly, underweight individuals, and those receiving insulin or insulin-like drugs. Fasting periods longer than 24 hours should be overseen by a healthcare practitioner. Proper intake of noncaloric fluids is crucial to ensure adequate hydration. Lastly, fasting may result in changes in circadian rhythms of endocrine and gastrointestinal systems.27,28