What is collagen?
The most abundant structural protein in the body, collagen is responsible for the structure of skin, connective tissue, tendons, bones, and teeth. The major collagen types are I, II, and III, which form the structural fibrils of tissues.1
| Download the PDF |
Function
As the main group of structural proteins of the extracellular matrix, collagen proteins form elastic molecular networks to strengthen tendons and elastic sheets that support skin and internal organs. They also function as biologically active components of tissues including skin, bone, and cartilage.2
Sources for supplementation
Collagen is extracted from animal byproducts, including both land
(e.g., bovine, porcine, and chicken) and marine sources, such as skin,
bones, connective tissue, fish heads, guts, fins, and scales.1,3 Marine
collagen, however, contains a lower amount of the amino acids proline
and hydroxyproline—resulting in lower denaturation temperatures
(lower melting point), which indicate their lower stability (weaker gel
strength).4 Additionally, biological properties of marine collagen can be
highly variable depending on biochemical composition.5
Collagen forms
Due to its highly preserved sequence and structure, collagen
and its derivatives (hydrolysates, peptides, and gelatin) are highly
biocompatible and safe.6-7
- Protein: Approximately 25-35% of all vertebrate proteins6 mainly
consist of three nearly identical α-chains of the amino acids glycine,
proline, and hydroxyproline. The unique amino acid composition and
structure confer collagen proteins with excellent stability.1 - Hydrolyzed: A result of the enzymatic breakdown (hydrolysis) of
collagen, the byproduct is extracted and dehydrated into a white
powder (hydrolyzed collagen or collagen hydrolysate) that is easier to
digest and use.1 - Peptides: These water-soluble peptide fragments (2-20 amino
acids) originate from the breakdown of the full-length collagen
protein. Due to their smaller size, collagen peptides show higher
bioavailability and are better absorbed into the bloodstream.8 - Gelatin: This denatured form of collagen is obtained by partial
hydrolysis of the protein. Depending on the pH, temperature, and
extraction process used, gelatin may vary in functional properties
despite having amino acid content similar to collagen.1
Research Highlights
- Collagen is the most abundant type of structural protein
in the body. - Supplement sources are extracted from animal byproducts.
- Marine collagen contains a lower amount of proline and
hydroxyproline as compared to bovine collagen—
resulting in lower stability and shelf-life. - Collagen protein and its derivatives (hydrolysates, peptides,
and gelatin) are highly biocompatible and safe. - Preclinical and clinical studies show that supplementation with
collagen may support skin, nail, bone, and joint health.
Health benefits
An increasing number of both animal and human studies have identified
a variety of bioactivities for collagen hydrolysates or peptides. Sustainable
sources, good bioavailability, and neutral taste render collagen hydrolysates
and peptides a promising approach for biomedical applications.
- Extracellular matrix and cartilage
- Preclinical studies show stimulation of chondrocytes to synthesize
extracellular cartilage matrix and changes in serum biomarkers
associated with increased collagen synthesis and decreased
extracellular matrix disruption.9-10 - Adults with mild knee osteoarthritis showed improved extracellular
matrix and cartilage support following daily supplementation with
10 g of collagen hydrolysate for 48 weeks.11
- Preclinical studies show stimulation of chondrocytes to synthesize
- Skin and nails
- Several clinical trials have highlighted the benefits of collagen peptides
on skin properties, such as hydration, elasticity, and reduction of wrinkles. - Collagen peptides can stimulate the growth of fibroblasts in the skin
and the synthesis of hyaluronic acid12 as well as improve skin barrier
dysfunction.13 - Daily consumption of 2.5 g of hydrolyzed collagen containing peptides
types I and III for 4 weeks increased skin elasticity in healthy female
subjects compared to placebo.14 - After 8 weeks of consuming 2.5 g per day of collagen peptides,
increased content of procollagen type I and elastin was observed as
compared to placebo-treated patients.15 - 2.5 g of collagen peptides consumed daily for 24 weeks showed
increased nail growth and improved brittle nails in conjunction with
decreased frequency of broken nails.16
- Several clinical trials have highlighted the benefits of collagen peptides
How Collagen Works in the Body
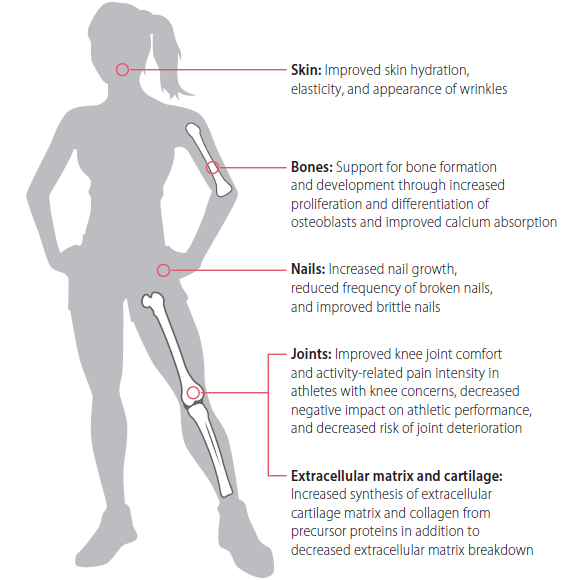
Joint and bone health
Collagen has been associated with improvements in joint and bone health.17
- Collagen peptides can stimulate the proliferation and differentiation
of osteoblasts and improve calcium absorption18 in addition to
up-regulating expression of bone-related genes in osteoblasts,19-20
suggesting that collagen supports bone formation and development.21 - 5 g of collagen peptides per day for 12 weeks improved activity-related
pain intensity in athletes with knee problems compared to placebotreated
subjects.22 - Osteoarthritis patients who supplemented daily with 10 g of collagen
hydrolysate for 24 weeks showed improvement in knee joint comfort.23 - Enhanced collagen synthesis as a result of daily supplementation with
10 g of collagen hydrolysate from 12 to 24 weeks may reduce the risk of
joint deterioration—indicating that the use of collagen supports joint
health and may reduce parameters that have a negative impact on
athletic performance (such as pain).24-25 - Postmenopausal women with age-related reduction in bone mineral
density supplemented with 5 g of collagen peptides per day for
12 months and showed increased bone mineral density and favorable
changes in bone markers compared to the control group.26
- Other applications
Collagen proteins are being considered in regenerative medicine for
tissue engineering and regeneration purposes, such as the production
of hydrogels or scaffold for wound healing. Collagen-based materials can
be used to prevent moisture and heat loss from wounded tissue, while
providing a barrier against microbial infiltration.27


