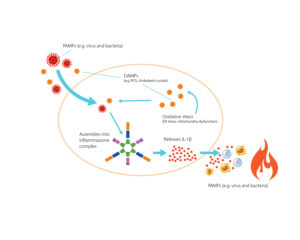
What is an inflammasome?
Inflammasomes are protein complexes, built by “ready-to-go” parts within a cell.1 Internal and external danger signals, such as an infection and cellular damage, can trigger inflammasome assembly in as little as 10 minutes.1,2 After activation, inflammasomes cause release of proinflammatory cytokines such as IL-1β and cascade to an orchestrated inflammatory cell death known as pyroptosis and downstream immune responses.1 Several inflammasomes exist, and NLRP3 is considered the most clinically relevant inflammasome.3
Why do we have inflammasomes?
Inflammasomes are part of the innate immune defenses and primarily function to keep infections from spreading.1
Are inflammasomes always helpful?
Inflammasome activation and pyroptosis are highly inflammatory, and the response can backfire, especially if not well-regulated.1 For example, NLRP3 inflammasome activity is responsible for acute respiratory distress syndrome (ARDS), seen secondary to pneumonia or sepsis.4 Inflammasome activation has been linked to diseases not usually associated with infections, such as Alzheimer’s, type 2 diabetes, autoimmune diseases, and atherosclerosis.2
Research Highlights
✓ Inflammasomes are the cellular machinery that drive inflammation
✓ Inflammasome activation amplifies inflammation
✓ Nutritional bioactives help regulate inflammasome activation
Nutritional bioactives that modulate inflammasome activation
Nutritional bioactives are compounds found in the diet that have biological effects. Certain nutritional bioactives have been studied for their specific action in certain physiological conditions.
Quercetin
Quercetin, a polyphenol found in many fruits and vegetables, has been shown to reduce the expression of NLRP3 inflammasome components and secretion of proinflammatory cytokines such as IL-1β.5 Quercetin suppresses the NF-κB pathway that also leads to reduced activation of NLRP3 inflammasome and productions of proinflammatory cytokines.6 Quercetin helps ameliorate mitochondrial dysfunction and endoplasmic reticulum stress, suppressing reactive oxygen species (ROS) formation in inflamed tissues.7
N-acetylcysteine (NAC)
NAC is a precursor in the production of glutathione, and supplementation can increase tissue glutathione levels in humans.8 In an experimental model of ROS-triggered NLRP3 expression, treatment with NAC suppressed the activation of the inflammasome.9 NAC regulates mRNA expression of lipopolysaccharide (LPS)-triggered NLRP3 inflammasome, lowering cytokines such as IL-1β.10
Zinc
Zinc is a vital nutrient for proper immune function. Zinc deficiency (especially common in older populations) in immune cells activates the NLRP3 inflammasome, induces IL-1β secretion, and contributes to inflammation.11 Zinc inhibits NLRP3 by activating the Nrf2 antioxidant pathway and reduces the production of ROS.12
Epigallocatechin-gallate (EGCG)
EGCG, a polyphenol commonly found in green tea, suppresses NLRP3 inflammasome mRNA, activation, and oxidative stress, reducing IL-1β expression in inflamed cells.7,13 EGCG activates Nrf2 antioxidant pathway, decreasing NLRP3 inflammasome activation.13 EGCG also inhibits NF-κB activation and reduces expression of multiple inflammatory signals such as TNFα, IL-6, iNOS, and MMPs.14
|
Nutritional bioactives influence inflammasome activation:
|
Vitamin C
Preclinical evidence shows vitamin C and quercetin together act as antioxidants and block a critical triggering event of the NLRP3 inflammasome.15 Vitamin C inhibits NLRP3 activation through scavenging mitochondrial ROS, rather than by inhibiting NF-κB.16
Vitamin D
Vitamin D has two unique pathways that regulate inflammasome activation.17 Vitamin D signaling through vitamin D receptor (VDR) binding is one mechanism.17 VDR can also directly bind to NLRP3 and prevent the inflammasome from assembling.17 Defects in this mechanism may be involved in the development of allergic diseases, as inflammasome activation is involved in T helper type 2 responses.18
Specialized pro-resolving mediators (SPMs)
SPMs, bioactives that can be found in specialized fractions of fish oil, promote inflammation resolution without causing immunosuppression.19 Supplementing SPMs enhances pathogen phagocytosis in immune cells.20
In an animal model SPM RvD2 blocks NLRP3 inflammasome activation, reduces IL-1β, and reprograms macrophages to a proresolving phenotype.21
In mice with lung infections, SPMs RvD1 and RvE1 increase bacterial clearance and shorten the time to inflammation resolution.22,23 SPM PD1 inhibits H1N1 and H5N1 influenza viral replication in human lung cells.24
Summary
A normal inflammatory response and activation of inflammasome protects the host from infection or prevents further tissue damage. However, prolonged dysregulated inflammasome activities have been associated with development of immune and metabolic disorders. Nutritional bioactives may provide a safer approach in addressing the harmful effects associated with dysregulated inflammation and inflammasome activation.
NAC is a precursor in the production of glutathione, and supplementation can increase tissue glutathione levels in humans.8 In an experimental model of ROS-triggered NLRP3 expression, treatment with NAC suppressed the activation of the inflammasome.9 NAC regulates mRNA expression of lipopolysaccharide (LPS)-triggered NLRP3 inflammasome, lowering cytokines such as IL-1β.10