by Anu Desai, PhD
Messenger RNA (mRNA) is a single-stranded molecule of genetic code found in all living cells. As a chemical intermediary, mRNA carries instructions from deoxyribonucleic acid (DNA) in our chromosomes to cellular machinery in the cytoplasm of the cell to make proteins.1
Over the past decade, research and technological innovations progressed rapidly in the field of vaccine development enabling mRNA as a promising therapeutic tool. There has been research on mRNA vaccines for infectious agents, such as Ebola, Zika virus, and influenza.1
Both Pfizer-BioNTech and Moderna mRNA vaccines carry information that tell our cells to make small fractions of the spike protein that is unique to severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), the virus that causes coronavirus disease 2019 (COVID-19). The spike protein is the part of the virus that enables it to enter our cells and cause infection.1
Since the instructions carried within the mRNA of the vaccines provide only enough information for our cells to make pieces of the spike protein, they do not pose any risk of infection.
1. Does COVID-19 mRNA vaccine change my DNA/affect my DNA?
No. This is because mRNA is different from DNA in that it is active only in the cytoplasm of the cell and cannot enter into the nucleus to interact with DNA to change the genetic code.2
2. How is the COVID-19 vaccine different from traditional vaccines?
Traditional vaccines contain either a weakened or an inactive form of a virus. The difference with mRNA vaccines is that they deliver mRNA which gives instructions to the cells to produce viral proteins. In the case of the COVID-19 vaccines, the mRNA gives instructions to the cells to produce pieces of the spike protein unique to SARS-CoV-2. Our immune cells recognize these spike protein pieces as foreign and initiate an immune response to develop antibodies, without posing any risk of infection. Thus, the body learns to build the defense it needs to protect against potential future infection of COVID-19.2
3. Can I get COVID-19 from taking mRNA vaccines?
No, the Pfizer-BioNTech and Moderna’s COVID-19 mRNA vaccines cannot give you COVID-19 since they do not contain SARS-CoV-2, the live virus that causes the illness.3,4
Reported side effects associated with Pfizer-BioNTech and Moderna COVID-19 vaccines are mild to moderate in intensity and include pain, redness, and swelling at injection site, tiredness, muscle pain, joint pain, headache, chills, and/or fever.3,4
4. If I get the vaccine, can I pass COVID-19 onto others?
COVID-19 mRNA vaccines can prevent the disease; however, both of the currently approved vaccines are about 95% effective.5 This means that if people are exposed to the virus after receiving one of the approved COVID-19 vaccines, there is still a chance that they could be infected and, therefore, could spread the virus to others. Even after vaccination, mask wearing, handwashing, and social distancing are important practices in controlling the spread of COVID-19.5
5. What are the ingredients in the mRNA vaccines?
The mRNA vaccines by Pfizer-BioNTech and Moderna include mRNA, lipids, salts, and sugar. The mRNA codes for the SARS-CoV2 spike protein and the lipids (fats) help protect the mRNA from breaking down before it gets into the cells. The added salts help maintain the pH of the vaccine so it is the same as that of the human body. Additionally, sugar helps keep the lipids from sticking to one another as well as to the vials that contain the vaccines.3,4
The Pfizer-BioNTech COVID-19 vaccine contains the following ingredients: mRNA, lipids ((4hydroxybutyl)azanediyl)bis(hexane-6,1-diyl)bis(2-hexyldecanoate), 2 [(polyethylene glycol)-2000]-N,N-ditetradecylacetamide, 1,2-Distearoyl-sn-glycero-3-phosphocholine, and cholesterol), potassium chloride, monobasic potassium phosphate, sodium chloride, dibasic sodium phosphate dihydrate, and sucrose.3
The Moderna COVID-19 vaccine contains the following ingredients: mRNA, lipids (SM-102, polyethylene glycol [PEG] 2000 dimyristoyl glycerol [DMG], cholesterol, and 1,2-distearoyl-sn-glycero-3-phosphocholine [DSPC]), tromethamine, tromethamine hydrochloride, acetic acid, sodium acetate, and sucrose. 4
6. Why is it necessary to get both doses of the vaccine?
It is not uncommon for vaccines to require two, three, or more doses in order to be most effective. Both the Pfizer-BioNTech and Moderna vaccines require two doses in order for them to work efficiently. The first dose will help prime the immune system to create a response against SARS-CoV-2, and the second shot helps boost that immune response. The effectiveness of the vaccine increases greatly after a second dose, and it provides longer-lasting immunity than a single dose.5 There are no long-term studies performed to demonstrate how long the immunity can last.
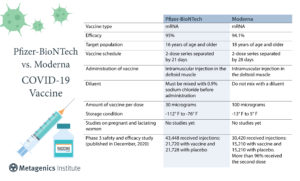
7. What are the differences between the Pfizer and Moderna vaccines?3-11
Further resources*
FDA COVID-19 Vaccine Information Page
NY Times Coronavirus Vaccine Tracker
*Metagenics Institute has no affiliation with these organizations.
Disclaimer: This information is current as of January 9, 2021. While this existing science is exciting and complex, every day brings new developments and discoveries that aren’t captured here because they haven’t happened yet!
Citations
- Different COVID-19 Vaccines. https://www.cdc.gov/coronavirus/2019-ncov/vaccines/different-vaccines.html. Updated December 28, 2020. Accessed January 7, 2021.
- Understanding and Explaining mRNA COVID-19 Vaccines. https://www.cdc.gov/vaccines/covid-19/hcp/mrna-vaccine-basics.html. Reviewed November 2020. Accessed January 7, 2021.
- Pfizer-BioNTech COVID-19 Vaccine EUA Fact Sheet for Recipients and caregivers. https://www.fda.gov/media/144414/download. Accessed January 7, 2021.
- Moderna COVID-19 Vaccine EUA Fact Sheet for Recipients and caregivers. https://www.fda.gov/media/144638/download. Accessed January 7, 2021.
- What to Expect after Getting a COVID-19 Vaccine. https://www.cdc.gov/coronavirus/2019-ncov/vaccines/expect/after.html#:~:text=With%20most%20COVID%2D19,get%20a%20second%20shot. Updated December 19, 2020. Accessed January 8, 2021.
- Frequently Asked Questions about COVID-19 Vaccination. https://www.cdc.gov/coronavirus/2019-ncov/vaccines/faq.html. Updated December 29, 2020. Accessed January 7, 2021.
- S. COVID-19 Vaccine Product Information. https://www.cdc.gov/vaccines/covid-19/info-by-product/index.html. Accessed January 7, 2021.
- Polack M et al. Safety and efficacy of the BNT162b2 mRNA Covid-19 vaccine. N Engl J Med. 2020;383(27):2603-2615.
- Rubin EJ and Longo DL. SARS-CoV-2 vaccination — an ounce (actually, much less) of prevention. N Engl J Med 2020;383:2677-2678.
- Baden LR et al. Efficacy and safety of the mRNA-1273 SARS-CoV-2 vaccine. N Engl J Med. 2020.
- Castells MC and Phillips EJ. Maintaining safety with SARS-CoV-2 vaccines. N Engl J Med. 2020.
Anu Desai, PhD is the Foundational Health Platform lead in the Department of R&D and has been with Metagenics for 15 years. She received her PhD in Nutritional Biochemistry and Life Sciences from the University of Hyderabad, India. At Metagenics, Dr. Desai has managed research programs and utilized state-of-the-art platform technologies to develop nutritional supplements and therapeutic medical food products for the maintenance and treatment of various diseases including arthritis, detox, and cardiometabolic and women’s health areas, which led her to publish several patents and research articles. She continues her work in helping people outside of Metagenics and dedicates much of her free time providing support to local homeless and women’s shelters.