
by Anu Desai, PhD
Pollution and toxic elements
Environmental pollution and toxins from plastics, pesticides, smoke, contaminated water, and food have the potential to greatly impact human health.1 These toxic loads cause inflammatory and autoimmune responses, including neurodegenerative conditions including Alzheimer’s disease (AD), Parkinson’s disease (PD), and multiple sclerosis (MS).2-4
More recently, some efforts have been made to minimize the exposures to pollutants in the long run, such as: using BPA-free plastics, attempting to reduce greenhouse gasses, and incorporating clean-energy technologies. 5 However, these efforts may not be good enough in controlling pollution in the short term. For example, recent fires from California to Washington State in 2020 caused dangerous levels of air pollution, which not only affect people with breathing sensitivities but also healthy people of all ages.6 Additionally, recent findings of microplastics in the Himalayan mountain range is a stark reminder of how pollution can spread to even the most remote communities. Contaminated drinking water in Flint, Michigan, is another example of environmental pollution causing developmental issues in children.7
Nrf-2 and detoxification
One way our bodies are equipped to handle biological/biochemical challenges is Nrf-2 transcriptional factor, which is involved in the regulation of antioxidant, anti-inflammatory, etc. properties. The NRF2/KEAP1 pathway is a key cellular defensive mechanism providing protection against environmental challenges caused by electrophiles, oxidants, and xenobiotics.8 Nrf-2 is a transcriptional factor; following its activation, Nrf-2 translocates to the nucleus and binds to genes containing antioxidant response elements. These genes are also involved in phase 1 and 2 detoxification mechanisms, among others. 9-10
Phase 2 enzymes are involved in cellular biotransformation of xenobiotics and endobiotics. Phase 2 traditionally refers to the enzymes catalyzing the conjugation reactions, such as glutathione S-transferase (GST), UDP-glucuronosyltransferase (UGT), N-acetyltransferase (NAT), and sulfotransferase (SULT).11 In addition, Nrf-2-dependent genes heme oxygenase-1 (HO-1) and NAD(P) H:quinone oxidoreductase type 1 (NQO1) are also involved in detoxification, though they are not directly involved in the biotransformation process. 12
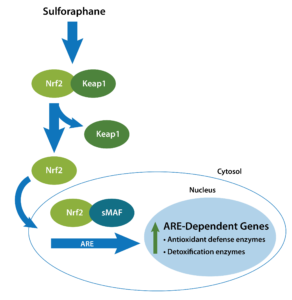
Nrf-2 modulates immune mechanisms
Increased Nrf-2 expression lowers the risk caused by diesel exhaust,13 and disruption of Nrf2 pathway enhances susceptibility to severe airway inflammation and asthma in mice.14 In humans, supplementation with sulforaphane (100mM), a potential activator of Nrf-2, improves the bronchoprotective response in asthmatics through Nrf-2 mediated gene pathways.12 In a different study, it was learned that consuming 200 g of broccoli sprouts (a rich source of sulforaphane) induced phase II enzyme expression in the airway, suggesting a potential role of Nrf-2 in reducing inflammation associated with an allergic response.15
Nrf-2 regulates viral replications
Recent research on antiviral programs indicates that Nrf-2 agonists 4-octyl-itaconate (4-OI) and dimethyl fumarate (DMF) inhibits replication of several viruses including SARS-CoV2, herpes simplex virus-1 and-2, vaccinia virus, and Zika virus through a type I interferon (IFN)-independent mechanism.16 There is growing evidence of a crosstalk between the Nrf2 and inflammasome pathways at different levels. For example, Nrf2-activating compounds inhibit inflammasome and consequently inflammation.17 These data indicate that Nrf-2 plays a role in the regulation of inflammasome and rejuvenation of immunity.
Science of sulforaphane
Among natural compounds, sulforaphane is the most potent molecule that activates Nrf-2-signaling mechanism. Glucoraphanin is a precursor to sulforaphane and is present in many cruciferous vegetables including broccoli, brussels sprouts, cabbage etc.18 Sulforaphane is produced when the enzyme myrosinase transforms glucoraphanin upon damage to the plant (or chewing), which allows the two compounds to mix and react. In addition, microorganisms within the gut can catalyze the hydrolysis of glucosinolates to sulforaphane.1
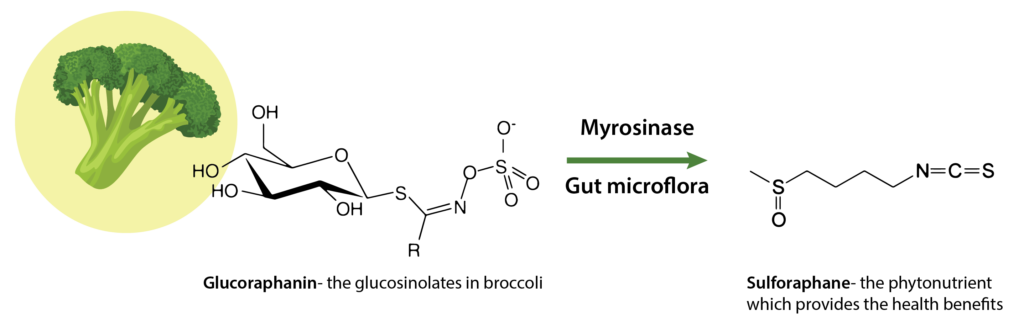
Due to the electrophilicity of the central carbon of the isothiocyanate (ITC) (-NCS) group, sulforaphane possesses high chemical reactivity.19 At high or neutral pH, sulforaphane is the primary product of glucoraphanin hydrolysis. In contrast, at acidic pH, or in the presence of cofactor epithiospecifier protein (ESP), the production of less active sulforaphane-nitrile will be favored. Heating broccoli sprouts to 60-700 C causes inactivation of ESP complex and favors a higher production of bioactive sulforaphane by the same enzyme myrosinase, which is resistant to heat inactivation at high temperature compared to ESP.20 So eating raw broccoli sprouts provides a much better source of glucosinolates, and therefore sulforaphane, compared to cooked broccoli sprouts.
Interestingly, in a double-blind placebo-controlled study, consumption of 100 g of broccoli sprouts for three days did not result in significant changes in pulmonary inflammation, oxidative stress, or clinical symptoms in adults with atopic asthma compared to alfalfa sprouts, likely due to the short duration of the intervention.21 It has been observed that glucoraphanin levels from diet directly correlated with the sulforaphane metabolites in the body.22 These studies suggests that consuming a glucoraphanin-rich diet for longer time at higher levels may be required to observe positive health benefits in clinical settings.
Sulforaphane in the diet & its bioavailability
Glucoraphanin levels in broccoli could be varied substantially dependent on genotype, growing conditions, and storage conditions, which makes it difficult to get consistent levels in the diet.23 Broccoli seeds and three-day-old sprouts contain 10 to 100 times more glucoraphanin, on a weight basis, than mature broccoli heads.23 In many clinical studies sulforaphane levels used ranged from 10-60 mg or equivalent to 25-150 mg of glucoraphanin.1
During the chewing process and in the presence of ESP, glucoraphanin converts to much less active sulforaphane-nitrile.20 Cooked broccoli florets or sprouts inactivates the myrosinase enzyme and produce 3-4 times less sulforaphane levels in the body.24 These studies point out that not only what part we eat, but also how we prepare it is important to get therapeutic doses of ITC for meaningful clinical outcomes.
Supplementation of sulforaphane
Delivering a therapeutic dose of sulforaphane in the diet has many challenges. Researchers are working to improve the stability of the sulforaphane. To date, no supplement is available in the market with sulforaphane that is stable at room temperature. Several studies indicate that active myrosinase enzyme is needed to convert glucoraphanin to sulforaphane. Compared to fresh broccoli, consuming heat-inactivated broccoli by cooking reduces the urinary metabolites of sulforaphane in humans.14 In another study, glucoraphanin supplementation along with active myrosinase enzyme improved the levels of ITC in the body.25 These led to the development of dietary supplements of glucoraphanin with active myrosinase that result in higher levels of sulforaphane yields.
Concluding remarks
Based on the clinical potential of sulforaphane and its versatility, eating a glucoraphanin-rich diet or taking supplements with glucoraphanin and active myrosinase could be helpful in reducing toxic burden in the body, leading to improved health outcomes.
Citations
- Houghton CA. Sulforaphane: its “coming of age” as a clinically relevant nutraceutical in the prevention and treatment of chronic disease. Oxid Med Cell Longev. 2019;2716870.
- de Figueiredo SM et al. The antioxidant properties of organosulfur compounds (sulforaphane). Recent Pat Endocr Metab Immune Drug Discov. 2015;9(1):24-39.
- Uddin MS et al. Emerging promise of sulforaphane-mediated Nrf2 signaling cascade against neurological disorders. Sci Total Environ. 2020;707:135624.
- Schepici G et al. Efficacy of sulforaphane in neurodegenerative diseases. Int J Mol Sci. 2020;21(22).
- Wazir U et al. Bisphenol A: a concise review of literature and a discussion of health and regulatory implications. In Vivo. 2019;33(5):1421-1423.
- Holm SM et al. Health effects of wildfire smoke in children and public health tools: a narrative review. J Expo Sci Environ Epidemiol. 2020;1-20.
- Gómez HF et al. Blood lead levels of children in Flint, Michigan: 2006-2016. J Pediatr. 2018;197:158-164.
- Bellezza I et al. Nrf2-Keap1 signaling in oxidative and reductive stress. Biochim Biophys Acta Mol Cell Res. 2018;1865(5):721-733.
- Hayes JD et al. The Nrf2 regulatory network provides an interface between redox and intermediary metabolism. Trends Biochem Sci. 2014;39(4):199-218.
- Cuadrado A et al. Transcription factor NRF2 as a therapeutic target for chronic diseases: a systems medicine approach. Pharmacol Rev. 2018;70(2):348-383.
- Boddupalli S et al. Induction of phase 2 antioxidant enzymes by broccoli sulforaphane: perspectives in maintaining the antioxidant activity of vitamins A, C, and E. Front Genet. 2012;3:7.
- Brown RH et al. Sulforaphane improves the bronchoprotective response in asthmatics through Nrf2-mediated gene pathways. Respir Res. 2015;16(1):106.
- Li YJ et al. Nrf2 lowers the risk of lung injury via modulating the airway innate immune response induced by diesel exhaust in mice. Biomedicines. 2020;8(10).
- Rangasamy T et al. Disruption of Nrf2 enhances susceptibility to severe airway inflammation and asthma in mice. J Exp Med. 2005;202(1):47-59.
- Riedl MA et al. Oral sulforaphane increases Phase II antioxidant enzymes in the human upper airway. Clin Immunol. 2009;130(3):244-251.
- Olagnier D et al. SARS-CoV2-mediated suppression of NRF2-signaling reveals potent antiviral and anti-inflammatory activity of 4-octyl-itaconate and dimethyl fumarate. Nat Commun. 2020;11(1):4938.
- Hennig P et al. The crosstalk between Nrf2 and inflammasomes. Int J Mol Sci. 2018;19(2).
- Fahey JW et al. Protection of humans by plant glucosinolates: efficiency of conversion of glucosinolates to isothiocyanates by the gastrointestinal microflora. Cancer Prev Res (Phila). 2012;5(4):603-611.
- Dinkova-Kostova AT et al. KEAP1 and done? Targeting the NRF2 pathway with sulforaphane. Trends Food Sci Technol. 2017;69(Pt B):257-269.
- Matusheski NV et al. Heating decreases epithiospecifier protein activity and increases sulforaphane formation in broccoli. 2004;65(9):1273-1281.
- Sudini K et al. A randomized controlled trial of the effect of broccoli sprouts on antioxidant gene expression and airway inflammation in asthmatics. J Allergy Clin Immunol Pract. 2016;4(5):932-940.
- Sivapalan T et al. Bioavailability of glucoraphanin and sulforaphane from high-glucoraphanin broccoli. Mol Nutr Food Res. 2018;62(18):e1700911.
- Farnham MW et al. Glucoraphanin level in broccoli seed is largely determined by genotype. Hortsci. 2005;40(1):50-53.
- Conaway CC et al. Disposition of glucosinolates and sulforaphane in humans after ingestion of steamed and fresh broccoli. Nutr Cancer. 2000;38(2):168-178.
- Fahey JW et al. Sulforaphane bioavailability from glucoraphanin-rich broccoli: control by active endogenous myrosinase. PLoS One. 2015;10(11):e0140963.
Anu Desai, PhD is the Foundational Health Platform lead in the Department of R&D and has been with Metagenics for 15 years. She received her PhD in Nutritional Biochemistry and Life Sciences from the University of Hyderabad, India. At Metagenics, Dr. Desai has managed research programs and utilized state-of-the-art platform technologies to develop nutritional supplements and therapeutic medical food formulas for the maintenance and treatment of various diseases including arthritis, detox, and cardiometabolic and women’s health, which led to the publish several patents and research articles. She continues her work in helping people outside of Metagenics and dedicates much of her free time to providing support to local homeless and women’s shelters.





I was diagnosed with multiple sclerosis ONE month after I turned FIFTY. My grandma is NINETY-SIX and had it since she was in her 20s. I have been on WORLDHERBSCLINIC MS HERBAL FORMULA, the first TWO MONTHS was daily and now I am on THREE times a week. It has made a tremendous difference for me. The fatigue never gets to me again. When I do too much, I don’t feel weak anymore.