by Mark Houston, MD and Sara Gottfried, MD
| Abstract. Cardiovascular disease, the leading cause of death in the United States, is not a single disease. It encompasses three different pathophysiological processes that involve the heart or the blood vessels. When considering whether omega-3 fatty acids have any benefit for our cardiovascular system, there is an enormous amount of misinformation in the news and reputable medical journals. Several articles state that omega-3 fatty acids have no role in cardiovascular disease, but unfortunately, researchers lump data from studies that investigate many different cardiovascular conditions for a single meta-analysis. When data from all those conditions are lumped together, it’s easy to miss effects for specific situations. This article will first review the flawed studies of meta-analyses and explain why they can lead to misinterpretation or misreporting. Then, we will examine studies that are properly conducted by focusing on solely coronary heart disease and how they reach a different conclusions regarding omega-3 fatty acids. Ultimately, we conclude that omega-3 fatty acids play a critical role both for risk reduction and for treatment in patients with coronary heart disease. |
Introduction
Cardiovascular disease is the leading cause of death in the United States. According to the Centers for Disease Control and Prevention, one person dies from it every 37 seconds.1 However, cardiovascular disease is a broad term that encompasses diseases that involve the heart (i.e., ‘cardio’) and diseases that involve the blood vessels (i.e., ‘vascular’). Diseases that involve the heart include, but are not limited to, coronary heart disease, angina, myocardial infarction (heart attack), heart failure, arrhythmia, cardiomyopathy, pulmonary heart disease, inflammatory heart disease, valvular heart disease, rheumatic heart disease, and congenital heart disease. Diseases that involve the blood vessels include peripheral artery disease, atherosclerosis, aneurysm, ischemic stroke, blood clotting disorders, renal artery disease, deep vein thrombosis, chronic venous insufficiency, and more. These conditions are diverse, and may involve different etiologies, diagnosis and treatment.
We require a new strategy that considers the system biology of cardiovascular disease within a precision and personalized medicine framework. Central to the new strategy of cardiovascular medicine is to consider the three finite responses of the blood vessel to an infinite number of insults. Those three responses include: inflammation, oxidative stress, and immune dysfunction.
Misinformation in the news and even reputable medical journals state that omega-3 fatty acids have no role in cardiovascular disease. However, researchers lump data from studies that investigate different cardiovascular conditions for meta-analysis. This article will review the flawed studies that have been reported and comb through the best science to put in perspective the omega-3 role in coronary heart disease—one of the most common types of cardiovascular disease.
A closer look at article title
When you read medical journal articles, one of the first things you likely do is look carefully at the title and, second, what was done related to study methodology. Here is an example of how the article title can be deceiving. One of the most publicized articles that came out asked, “What is the role of omega-3 fatty acids in cardiovascular disease?”2 Now, what do you think of when you hear cardiovascular disease? Well, the article brings up the following: (1) heart attack, (2) stroke, (3) total mortality, (4) coronary heart disease, (5) angina, (6) bypass graft, (7) needs for stents, (8) hospitalizations from heart disease, and (9) several others.2 In other words, it included everything you could think of that was called cardiovascular disease.
What if the title had been changed to “What is the role of omega-3 fatty acids in coronary heart disease?” Instead of thinking all the conditions listed above, you would look specifically in the article for what happened with coronary heart disease, and you would get a very different story. This is because when data from all those conditions are lumped together, it’s easy to miss effects for specific situations. When you do the net result, it comes out as a null, and that’s how this can lead to misinterpretation or misreporting.
Deciphering the nature of meta-analysis
Let’s begin with three relevant journal articles that recently came out. The first one is the one I mentioned a couple of paragraphs earlier, titled ‘Omega-3 Polyunsaturated Fatty Acid (Fish Oil) Supplementation and the Prevention of Clinical Cardiovascular Disease: A Science Advisory From the American Heart Association’ published in Circulation.2 The second one is titled ‘Associations of Omega-3 Fatty Acid Supplement Use With Cardiovascular Disease Risks: Meta-analysis of 10 Trials Involving 77917 Individuals’ published in JAMA Cardiology.3 The third one is titled ‘Omega-3 Fatty Acids for the Primary and Secondary Prevention of Cardiovascular Disease’ published in Cochrane Database of Systematic Reviews.4
These three journal articles that based on meta-analyses basically said, “Quit using omega-3 fatty acids; they don’t work for cardiovascular disease,”2-4 but none of them said it doesn’t work for coronary heart disease. That’s where we want to emphasize the nonreality of the meta-analyses when they were inappropriately conducted, how they could be flawed, how they could misinterpret or misreport their own data, and how those biased findings were then picked up by the news media and other journals, turning them into sensational headlines. Almost overnight through word of mouth, providers were advised to stop using omega-3 fatty acids.
On the JAMA Cardiology meta-analysis
We want to convince you that omega-3 fatty acids are very important and indicated in the prevention and treatment of coronary heart disease and myocardial infarction (most myocardial infarctions occur due to coronary heart disease). Let’s take one of the studies, published in one of the most reputable journals in the United States, Journal of the American Medical Association (JAMA) Cardiology.3 It was a meta-analysis that included nearly 78,000 participants from 10 randomized trials. That is a reasonable number of patients in a reasonable number of studies. The problem was, the studies were poor and highly biased in their selection. They really didn’t meet the criteria for a good clinical trial to be put into a meta-analysis.
Analysis and meta-analysis are like and physics and metaphysics. You’ve got to be careful about what a meta-analysis is all about, and if you put one or two bad studies in with 10 or 12 good studies, it skews the data, and that’s exactly what happened in the JAMA meta-analysis. They also didn’t even look at the dosing required, and the studies weren’t long enough to evaluate whether or not patients had benefited from omega-3 fatty acids. You’ve got to have a large number of patients, you’ve got to study them for at least five years, and you’ve got to look at a dose response.
If you’re giving 200 mg versus 4,000 mg of omega-3 fatty acids, the outcomes are going to be very different. You can’t make a judgment call unless you look at a dose-response curve or dose-response analysis. JAMA didn’t do that, and it reported that omega-3 fatty acids had no role in the treatment of coronary heart disease and cardiovascular disease. However, when you look at its data and pull out just the coronary heart disease and myocardial infarction data, it turns out it actually did have a response; 7% reduction in coronary heart disease, morbidity, and mortality, and myocardial infarction in the JAMA trial, not reported in the abstract, not reported in the conclusion, but actually in the results and reported by the authors.3 That’s what happens when you lump the data. That’s the JAMA story, which was misleading and incorrect in its analysis, but in the critical analysis, even JAMA showed a reduction in coronary heart disease and myocardial infarction, and it was dose-related; the higher the dose, the better the patient did.
On the Cochrane meta-analysis
The second article, the Cochrane analysis, was a much larger study that included 79 randomized controlled trials involving more than 112,000 participants.4 Cochrane has always had a good reputation, good statistics, but again, it’s a meta-analysis, meaning it looked at the clinical trials, put them all together, tried to determine what the theory is based on different types of studies, different populations, different duration, different doses.
Cochrane concluded that omega-3 fatty acids had no role whatsoever in cardiovascular disease. Once again if you look at the title of the Cochrane paper, it is cardiovascular disease, it did not say the role in coronary heart disease. A closer look through the Cochrane analysis—more than 500 pages—a careful reader may find something disturbing: the authors did not report the results correctly. In the team’s analysis, cardiovascular disease was defined not just by coronary heart disease or myocardial infarction; it also included congenital heart disease, rheumatic heart disease, cardiomyopathy—conditions that are not even related to coronary heart disease. Again, they lumped the data and reported on cardiovascular disease.4
If a reader focuses on data related to just coronary heart disease and myocardial infarction—the number one reason people die in the United States related to heart disease—a totally different story emerges. There was a 7% reduction in coronary heart disease and myocardial infarction. Further, there was information on dose response. In the low-dose group there was still a reduction of about 6-7% in coronary heart disease; but in the high-dose groups (taking over 4,000 mg a day) there was a 68% decrease in coronary heart disease and myocardial infarction. That did not get reported by the Cochrane authors; they totally ignored it and left it out. They also ignored the fact that both primary and secondary prevention were reduced in those patients related to coronary heart disease. When you look at all that, you realize those two studies, which reported negative results, were actually positive for omega-3 fatty acids related to coronary heart disease and myocardial infarction.
Two positive lessons
In contrast to the two meta-analyses mentioned above, some studies were conducted without lumping data from too many diverse disease conditions leading to data misinterpretation. Let’s have a closer look at these examples.
On the Mayo Clinic Proceedings meta-analysis
The first example is an article titled ‘A Meta-Analysis of Randomized Controlled Trials and Prospective Cohort Studies of Eicosapentaenoic and Docosahexaenoic Long-Chain Omega-3 Fatty Acids and Coronary Heart Disease Risk’ published in Mayo Clinic Proceedings.5 Their first analysis included 18 randomized controlled trials involving 93,000 participants, with the omega-3 dose ranging from 380 mg/day to 5,040 mg/day and the study duration lasting 6 months to 7 years. Their second analysis included 17 prospective cohort studies involving 732,000 subjects, with the study follow-up ranging from 6 years to 40 years.
Findings from both types of studies are not only important, they complement one another.6 Randomized controlled trials are usually shorter in duration due to logistics, and tend to involve subjects with more established disease or with higher disease risks, but have a great advantage in controlling potential confounders through randomization. Prospective cohort studies, on the other hand, are able to follow larger groups of participants who are healthy at baseline and evaluate them over longer follow-up periods in a more realistic setting and environment.
By having analysis from both randomized controlled trials and prospective cohort studies, this Mayo study was the most comprehensive of all the meta-analyses in evaluating the role of omega-3 fatty acids. It also powered better. There were 825,000 patients. It dwarfed the JAMA and Cochrane studies (the JAMA study had 78,000 patients, Cochrane had 112,000). Importantly, the Mayo study has the best meta-analysis ever reported related to its title, which is the role of omega-3 fatty acids in coronary heart disease. Its study inclusion criteria were specific; for coronary heart disease only.5
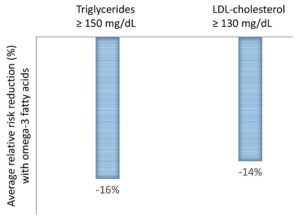
This Mayo analysis found a major reduction in coronary heart disease and all the different subsets, so anywhere from 7-25% reduction, depending on the patient population, the dose, and the duration of the trial. They also found that if your triglycerides were over 150 mg/dL you were particularly likely to get a response related to omega-3 fatty acids and myocardial infarction (a 16% risk reduction), and if your LDL cholesterol was over 130 mg/dL, you had a great response to omega-3 fatty acids related to coronary heart disease and myocardial infarction (a 14% risk reduction; Figure 1).5
Figure 1. Omega-3 fatty acid supplementation reduced coronary heart disease risk compared with placebo in at-risk populations (those with elevated triglycerides or LDL-cholesterol), according to the most comprehensive quantitative assessment by the Mayo Clinic.5
The lipid levels were not even looked at in the JAMA or the Cochrane analysis, but they were looked at as a response rate in the Mayo Clinic Proceedings analysis. There are clearly subsets of patients that had the greatest benefit, but even overall, taking the entire population, the omega-3 fatty acids showed a clear dose response.
On the NEJM REDUCE-IT trial analysis
The second example is an article titled ‘Cardiovascular Risk Reduction with Icosapent Ethyl for Hypertriglyceridemia’ published in New England Journal of Medicine (NEJM) in 2019.7 This is a prospective randomized clinical trial known as the Reduction of Cardiovascular Events with Icosapent Ethyl-Intervention Trial, or REDUCE-IT. The trial looked at a specific high-risk patient group—those who had heart disease, like coronary heart disease or myocardial infarction, or those that with type 2 diabetes and an additional coronary heart disease risk factor, both men and women usually in their 40s to 50s or older. These were patients who had been receiving statin therapy but still had substantial residual risks, including fasting triglyceride level of 135-499 mg/dL and LDL-cholesterol level of 41-100 mg/dL. Patients were randomly assigned to receive 2 g of eicosapentaenoic acid (EPA) ethyl ester twice daily (total daily dose of 4 g) or placebo for a median of 4.9 years.
The results at the end of the trial were very clear. Among at-risk patients who were receiving statin therapy, the rates of coronary heart disease-related primary and secondary end points were significantly lower in the omega-3 group than in the placebo group. Specifically:7
- 25% reduction in cardiovascular death or nonfatal myocardial infarction
- 31% reduction in fatal or nonfatal myocardial infarction
- 35% reduction in urgent or emergency revascularization
- 20% reduction in cardiovascular death
- 32% reduction in hospitalization for unstable angina
- 28% reduction in fatal or nonfatal stroke
When the researchers looked at the results in prespecified subgroups such as by sex, race, age, region, baseline lipids, and baseline high-sensitivity C-reactive protein, the benefits of the omega-3 fatty acid were consistent.
Conclusion
The data are in. We believe that based on the best data omega-3 fatty acids should be used both for risk reduction and for treatment in patients who have coronary heart disease and previous heart attack. Additionally, patients with triglycerides over 150 mg/dL, patients with an LDL-cholesterol level over 140 mg/dL, and patients with type 2 diabetes will benefit from taking omega-3 fatty acids, and higher doses are necessary to get the greatest benefit. You can give smaller doses and see improvement in outcomes, but it seems that 4,000 mg a day gave the highest benefit when we aggregate the clinical studies and all the meta-analyses.
Bottom line: get a high-quality omega-3 fatty acid supplement that is balanced, that has EPA, docosahexaenoic acid (DHA), gamma-linolenic acid (GLA), and gamma-delta tocopherols that allow the best absorption, and optimal clinical deposition in the cell membranes without oxidation. Based on the latest data, your patients will get the best results in reducing cardiovascular and coronary heart disease.
Citations
- Heron M. Deaths: Leading Causes for 2017. Natl Vital Stat Rep. 2019;68:1-77.
- Siscovick DS et al. Omega-3 polyunsaturated fatty acid (fish oil) supplementation and the prevention of clinical cardiovascular disease: a science advisory from the American Heart Association. Circulation. 2017;135:e867-e884.
- Aung T et al. Associations of omega-3 fatty acid supplement use with cardiovascular disease risks: meta-analysis of 10 trials involving 77917 individuals. JAMA Cardiol. 2018;3:225-234.
- Abdelhamid AS et al. Omega-3 fatty acids for the primary and secondary prevention of cardiovascular disease. Cochrane Database Syst Rev. 2018;11:CD003177.
- Alexander DD et al. A meta-analysis of randomized controlled trials and prospective cohort studies of eicosapentaenoic and docosahexaenoic long-chain omega-3 fatty acids and coronary heart disease risk. Mayo Clin Proc. 2017;92:15-29.
- Sorensen HT et al. Beyond randomized controlled trials: a critical comparison of trials with nonrandomized studies. Hepatology. 2006;44:1075-1082.
- Bhatt DL et al. Cardiovascular risk reduction with icosapent ethyl for hypertriglyceridemia. N Engl J Med. 2019;380:11-22.
Mark Houston, MD, MS, FAHA, FASH, DABC is a highly recognized cardiovascular and hypertension specialist, serving as a physician at St. Thomas West Hospital in Nashville, where he holds several leadership positions including Director of the Hypertension Institute and Vascular Biology, Medical Director of Clinical Research, and Section Chief for the Division of Nutrition. Dr. Houston cofounded the Hypertension Institute, which is one of the leading institutes in the US for hypertension treatment. An American Society of Hypertension (ASH) specialist and Diplomate of the American Board of Cardiology, Dr. Houston is also a Fellow of the American Society of Hypertension (FASH), the American Heart Association (FAHA), Internal Medicine (ABIM), American College of Nutrition (FACN) and Anti-Aging Medicine (ABAARM). Dr. Houston received his medical degree from Vanderbilt University. He also obtained two master’s degrees, in Human Nutrition and Metabolic Medicine and Human Nutrition, from the University of Bridgeport and University of South Florida, respectively.
Sara Gottfried, MD is a board-certified gynecologist and physician scientist. She graduated from Harvard Medical School and the Massachusetts Institute of Technology and completed residency at the University of California at San Francisco. Over the past two decades, Dr. Gottfried has seen more than 25,000 patients and specializes in identifying the underlying cause of her patients’ conditions to achieve true and lasting health transformations, not just symptom management.
Dr. Gottfried is a global keynote speaker who practices evidence-based integrative, precision, and Functional Medicine. She recently published a new book, Brain Body Diet and has also authored three New York Times bestselling books: The Hormone Cure, The Hormone Reset Diet, and Younger.